Az egységes szabályozási követelmények világszintű térnyerése egyre inkább megköveteli olyan orvostechnikai szabvány kidolgozását, amelyet az orvostechnikai eszköz gyártók teljes köre alkalmazni tud, és garantálja a termékek biztonságát és teljesítő képességét.
Az orvosi eszközökről szóló rendeletekkel (2017/745 (EU) és 2017/746 (EU) rendeletek) összhangban elvárás az orvostechnikai eszközök gyártóitól, hogy hatékony minőségbiztosítási rendszert vezessenek be és tartsanak fenn.
Az MSZ EN ISO 13485 alkalmazása az orvosi eszköz gyártója számára önkéntes, ugyanakkor az orvostechnikai eszköz piac jelentős részén kikerülhetetlen elvárás a minőségbiztosítási rendszer meglétének igazolására.
Az MSZ EN ISO 13485-ös szabvány leírja az orvostechnikai eszköz gyártója számára a minőségirányítási rendszer lényegi követelményeit.
A szabvány teljesítésével a szervezet be tudja bizonyítani, hogy folyamatosan képes a jogszabályi követelménynek eleget tevő, a szándékolt célt teljesítő orvosi eszközök előállítására és a kapcsolódó szolgáltatások biztosítására.
Az orvostechnikai eszköz-gyártók beszállítói számára sok esetben MSZ EN ISO 13485 szabványnak való megfelelés az egyetlen lehetőség a vonatkozó követelmények teljesítésének igazolására.
Az EN ISO 13485 tanúsítás nemcsak az európai piacon, hanem bárhol a világon alapvető jelentőségű.
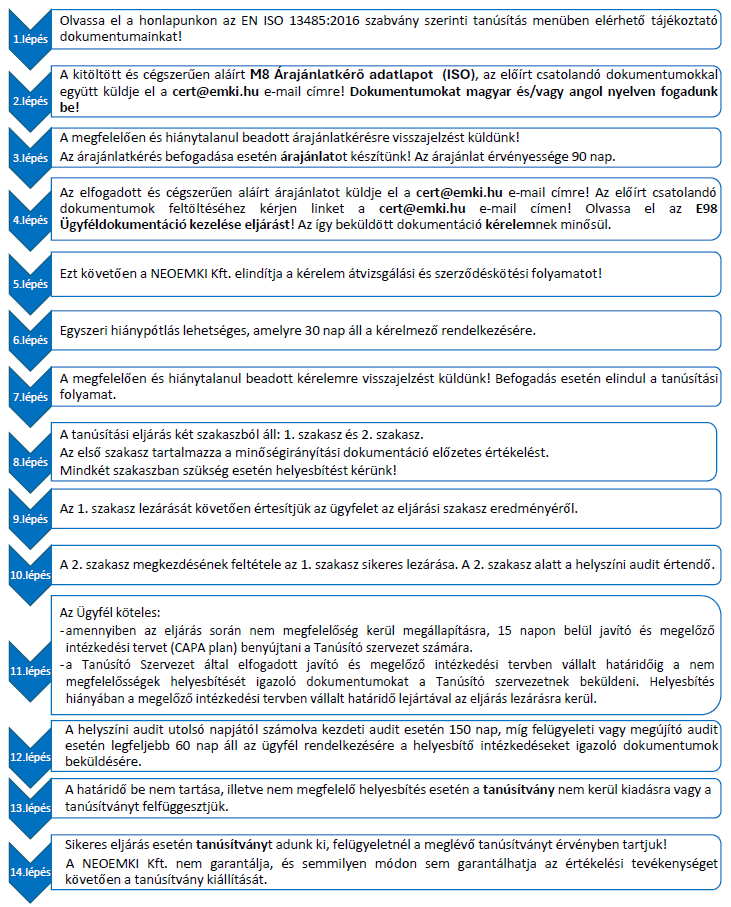
Cégünknél minőségirányítási rendszer tanúsítása az ajánlatkérő adatlap kitöltésével kezdeményezhető.
A tanúsítás menete